
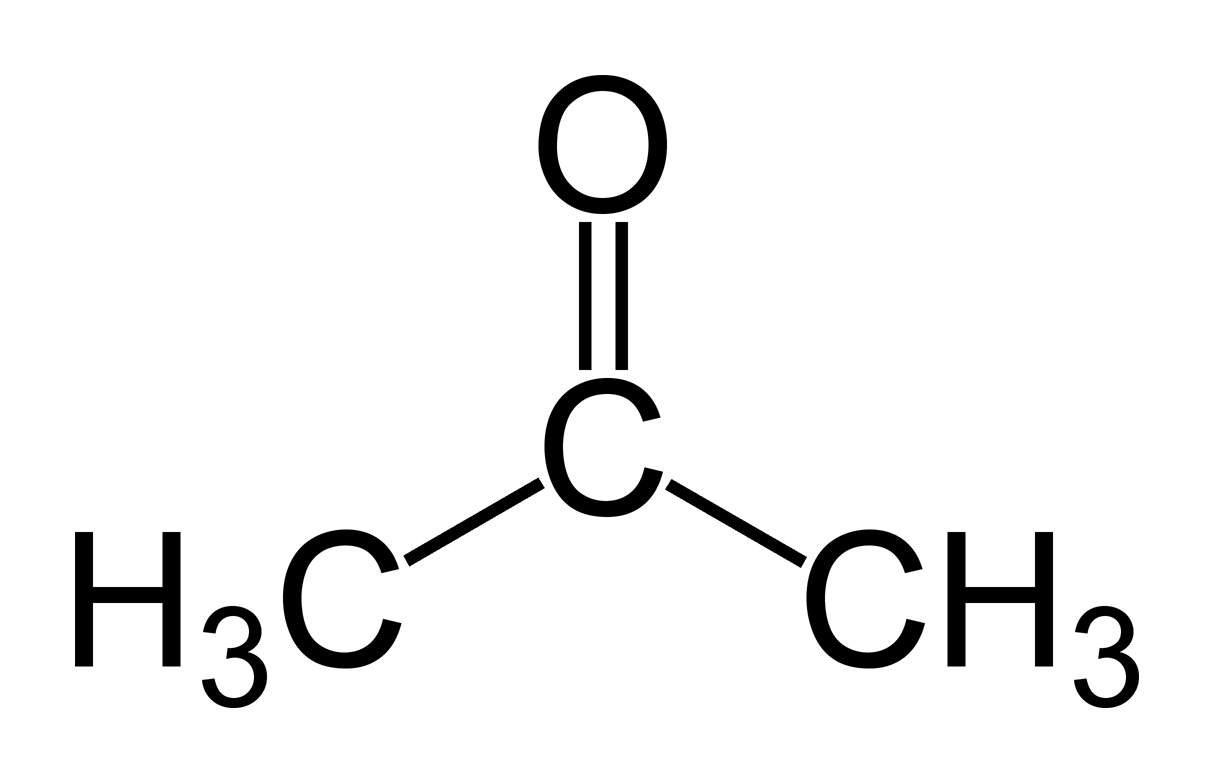
Propanone (Acetone)
Acetone contains a polar C=O double bond oriented at about 120° to two methyl groups with nonpolar C-H bonds. The C-O bond dipole therefore corresponds to the molecular dipole, which should result in both a rather large dipole moment and a high boiling point. Thus we predict the following order of boiling points:

In this video we are going to determine the polarity of CH3OH molecule
Propanone is very soluble in water because а. it is non-polar b. the dipole-dipole interactions are weak between propane and water С. it can form hydrogen bond with water molecule d. water is a polar solvent Chemistry: The Molecular Science 5th Edition ISBN: 9781285199047 Author: John W. Moore, Conrad L. Stanitski
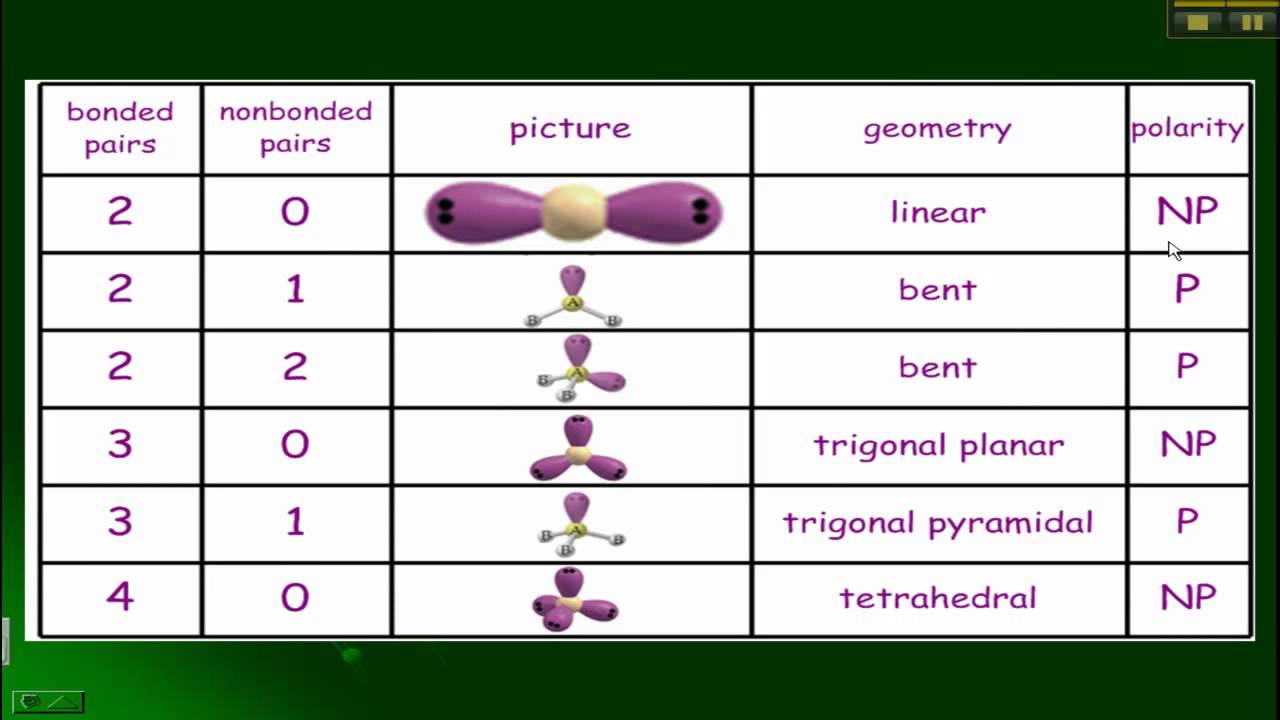
VSEPR, Polarity, and Bonds YouTube
The Rf value varies depending on the solvent used, but the general order of the pigments (from the highest to the lowest Rf value) usually remains the same, because the nonpolar compounds move further than the polar compounds. Rf values for various pigments (using hexane, acetone and trichloromethane (3:1:1) for the solvent) are shown in table 1.

Lewis Structure Of 1 Propanol
However, acetone is still considered a polar aprotic solvent, despite the fact that it is relatively acidic, and not significantly less acidic than alcohols. Then again, acetone (and other carbonyl containing solvents) are, indeed, poor solvents when using strong bases due to their relatively high acidity.

Découvrir 117+ imagen propanone formule fr.thptnganamst.edu.vn
- Techiescientist Is Acetone Polar or Nonpolar? Acetone is an organic compound with its chemical formula (CH3)2CO. It is classified as the simplest ketone. It exists as a colorless volatile liquid. It has a characteristic odor and flammable in nature. Many students may have a question regarding whether acetone is polar or not.
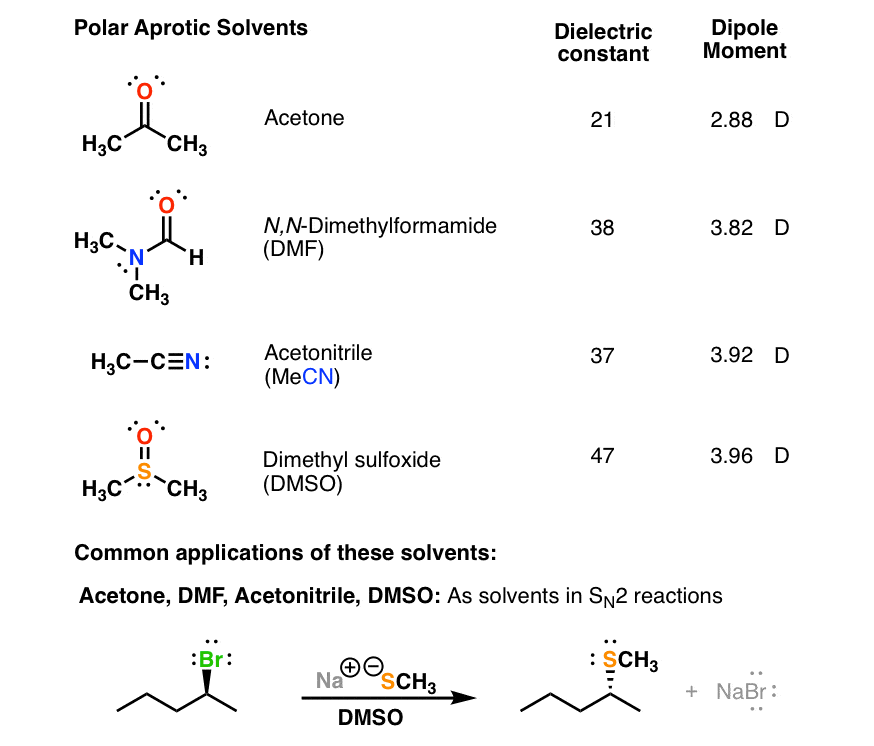
Polar Protic? Polar Aprotic? Nonpolar? All About Solvents
Common solvents arranged from the least polar to the most polar. Solvent Relative Polarity; hexane 0.009 p-xylene 0.074 toluene 0.099 benzene 0.111 ether 0.117 methyl t-butyl ether (MTBE). acetone 0.355 dimethylformamide (DMF) 0.386 t-butyl alcohol 0.389 sulfolane 0.41 dimethylsulfoxide (DMSO) 0.444 acetonitrile 0.46 nitromethane 0.481.
[Solved] 8. (5 pts. ) We understand phenomena such as solubility in
Propanone is SLIGHTLY POLAR and so can dissolve other polar substances-Propanone is LESS POLAR than water-this allows a RESOLUTION resolution between the pigment and the TLC plate as the pigments will dissolve in the propanone.
2Propanon
Propanone is normally written CH 3 COCH 3. Notice the need for numbering in the longer ketones. In pentanone, the carbonyl group could be in the middle of the chain or next to the end - giving either pentan-3-one or pentan-2-one.. Both aldehydes and ketones are polar molecules because of the presence of the carbon-oxygen double bond. As well.
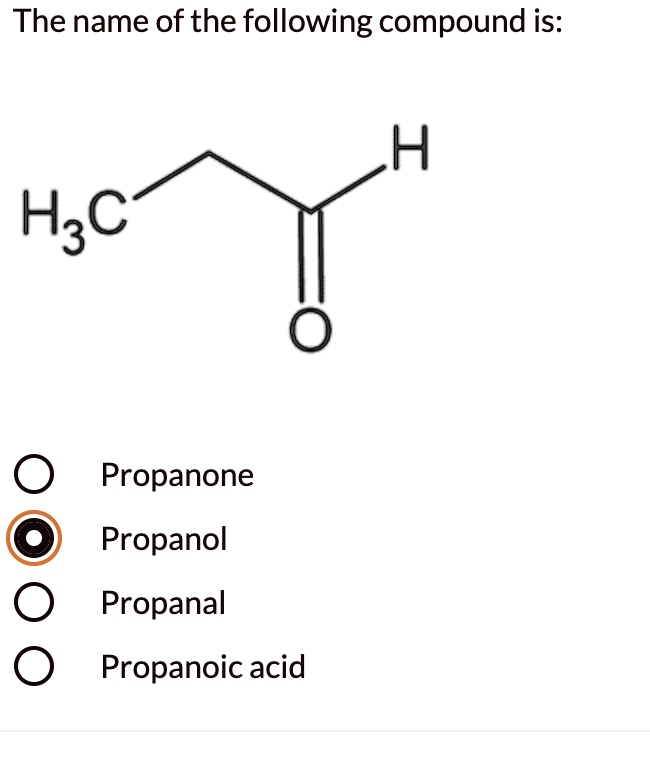
SOLVED The name of the following compound is H3C Propanone Propanol
This compound is also referred to as propanone or, in some texts as Propan-2-one. Acetone is considered to be the simplest form of Ketone. This compound is colorless, flammable, and is pungent smelling. It boils at temperatures of approximately 56° C.

Ppt Polar Bonds And Molecules Powerpoint Presentation, Free Download 587
217-218 °C Alfa Aesar: 218 °C Food and Agriculture Organization of the United Nations 1-Phenyl-1-propanone: 218 °C OU Chemical Safety Data (No longer updated) More details: 217-218 °C Alfa Aesar A15140: 218 °C Oakwood: 8 °C / 84 mmHg (68.4982 °C / 760 mmHg) FooDB FDB010567 218 °C Sigma-Aldrich SIAL-61074: 218 °C Oakwood 094675

Ch3Oh Lewis Structure Geometry Hybridization And Polarity itechguides
∙ 11y ago Study now See answer (1) Best Answer Copy Propane itself is non polar, but the presence of the ketone group (C=O) in propanone makes it a polar molecule (oxygen has partial -ve.

DP Chemistry Iodination of propanone
The carbonyl group is rather polar, however, since the difference between the electronegativities of carbon (2.5) and oxygen (3.5) is rather large, and there are usually no other dipoles in an aldehyde or ketone molecule to cancel the effect of C==O.. Like other ketones, acetone is mainly useful as a solvent, and you may have used it for.
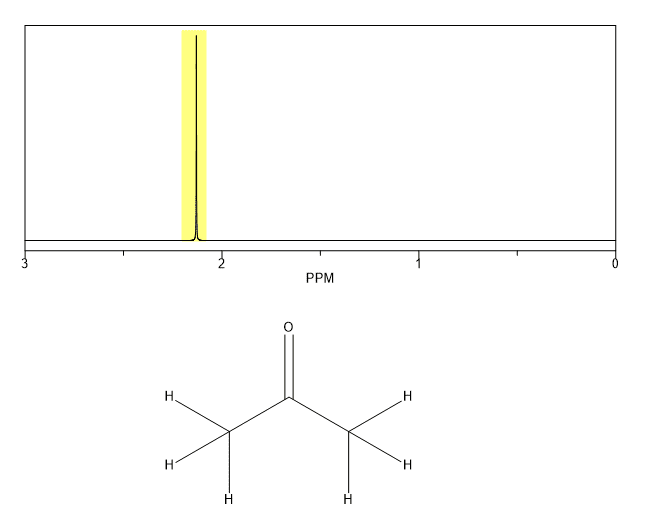
Propanone Proton NMR Equivalent Protons
Unlike polar bonds, non-polar bonds share electrons equally. A bond between two atoms or more atoms is non-polar if the atoms have the same electronegativity or a difference in electronegativities that is less than 0.4. An example of a non-polar bond is the bond in chlorine. Chlorine contains two chlorine atoms.
GCSE Chemistry C3 Organic Chemistry Revision Cards in GCSE Chemistry
Propanal, also known as propionaldehyde, is an organic compound with the chemical formula C3H6O. It is a colorless liquid with a pungent odor and is commonly used in the production of plastics, rubber, and various chemicals. Here are some examples of how to use propanal in a sentence: The lab technician synthesized propanal for the experiment.

What Is The Molecular Formula Of Propanone
Acetone ( 2-propanone or dimethyl ketone) is an organic compound with the formula (CH3)2CO. [22] It is the simplest and smallest ketone ( >C=O ). It is a colorless, highly volatile and flammable liquid with a characteristic pungent odor. Acetone is miscible with water and serves as an important organic solvent in industry, home, and laboratory.

Formula Estructural Del Propano Acido
Take acetone (C 3 H 6 O) for example. Acetone is polar because there is a partial negative charge on the oxygen and a partial positive charge on the rest of the molecule. This is an aprotic solvent because the highly electronegative atom, oxygen, is not bonded to a hydrogen atom. The hydrogen atoms are bonded to the carbon instead, which is a.